Research topics
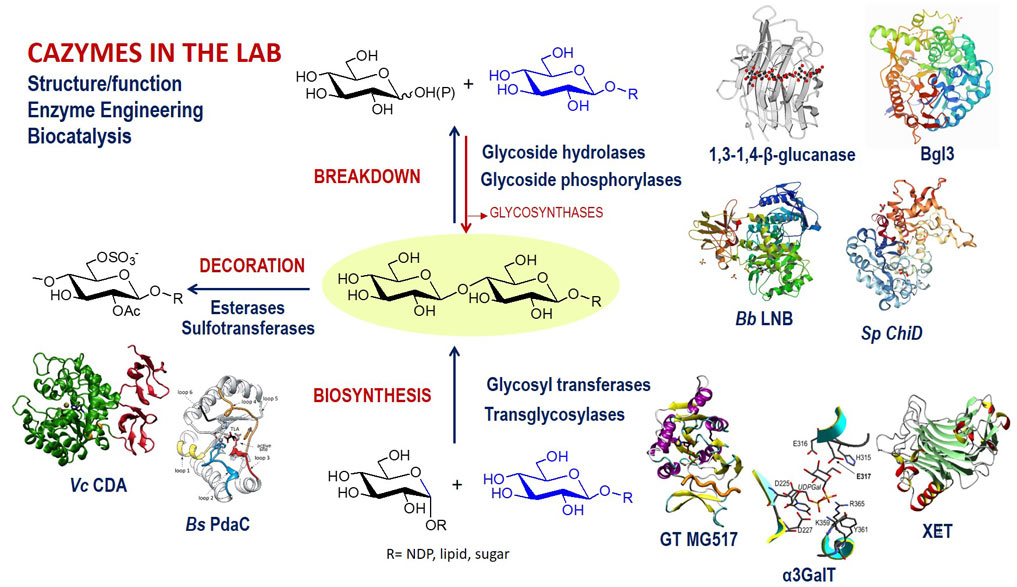
STRUCTURE/FUNCTION AND ENGINEERING OF CARBOHYDRATE ACTIVE ENZYMES. Applications in Biocatalysis.
Targeting glycosidases, glycosyltransferases and carbohydrate esterases, we study the mechanistic aspects of enzyme catalysis and the structural determinants of substrate specificity. Protein engineering on these enzyme families, either by knowledge-based design or directed evolution strategies enables their applications in biocatalysis. Main topics include: a) De-N-acetylation of structural polysaccharides by peptidoglycan and chitin deacetylases. b) Glycosynthesis by engineered glycosyl hydrolases as glycosynthases and transglycosidases. c) Structure-function of glycosyltransferases as therapeutic targets.
BIOINFORMATICS AND MOLECULAR MODELING IN PROTEIN STRUCTURE, FUNCTION, AND ENGINEERING.
Bioinformatics and computational simulations are key enabling technologies in protein engineering programs, aiming at the design and prediction of structure-function relationships. Our work is addressed by three main computational approaches: a) Bioinformatics: genome-wide search for novel functional enzymes or medical targets, and advanced methodologies for massive (big data) sequence and structure analysis of protein databases to infer function and evolutionary relationships; b) Molecular modeling: molecular dynamics and ligand docking of protein structures to address fundamental concepts of protein folding, reactivity, specificity and stability; c) Development and application of computational tools to assist experimental protein engineering campaigns (directed evolution) aimed at designing efficient biocatalysts.
METABOLIC ENGINEERING OF BACTERIA AND YEAST FOR THE PRODUCTION OF GLYCOLIPIDS
Progress in synthetic and systems biology has empowered metabolic engineering with tools and strategies to rewire the metabolism of microorganisms to optimize the flux to specific products of interest. We use E. coli (prokaryotic model) and S. cerevisiae (eukaryotic model) as metabolic engineering platforms targeting glycolipids as biomolecules with a broad range of applications: glycoglycerolipids as biosurfactans for drug delivery systems, and glycosphingolipids as immunomodulators.
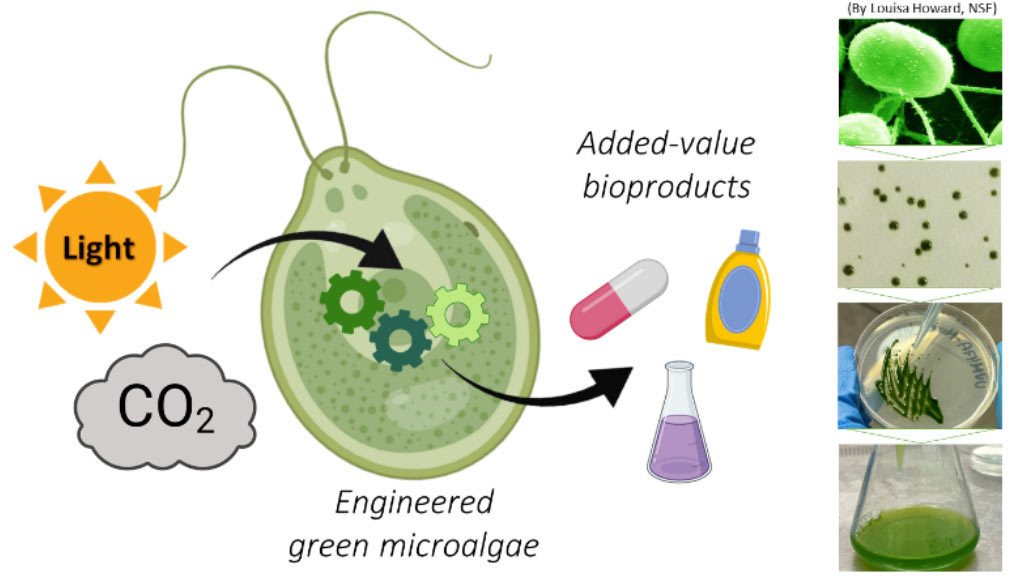
SYNTHETIC BIOLOGY AND MOLECULAR BIOTECHNOLOGY IN GREEN MICROALGAE
Microalgae are unicellular organisms that are emerging as important hosts for sustainable industrial biotechnology, as they can use photosynthetic light to efficiently transform CO2 into bioproducts with added value. Using the microalgae Chlamydomonas reinhardtii as a green cell chassis for synthetic biology, we aim at developing: (a) a metabolic engineering platform for the sustainable production of valuable glycolipids such as rhamnolipid biosurfactants; (b) an optogenetic module for the light-controlled expression of genes…
Construction of synthetic green microalgae for the sustainable production of added-value bioproducts.
BIOPROCESS TECHNOLOGY FOR THE PRODUCTION OF RECOMBINANT PROTEINS AND HIGH VALUE METABOLITES
Microbial cell factories are commonly used in industrial biotechnology to produce a broad diversity of compounds. Once the production microorganism has been developed (by genetic and metabolic engineering), upstream and downstream bioprocess technologies are essential to make production viable in terms of productivity and costs. Culture media design and optimization, and bioreactor operation conditions are central for high productivity in upstream processing. Our research focusses on the utilization of alternative non-food feedstocks for the production of high value compound such as recombinant proteins, vitamins and glycolipids